The information displayed in the AIM should not be reported as representing the official views of the OECD or of its member countries.
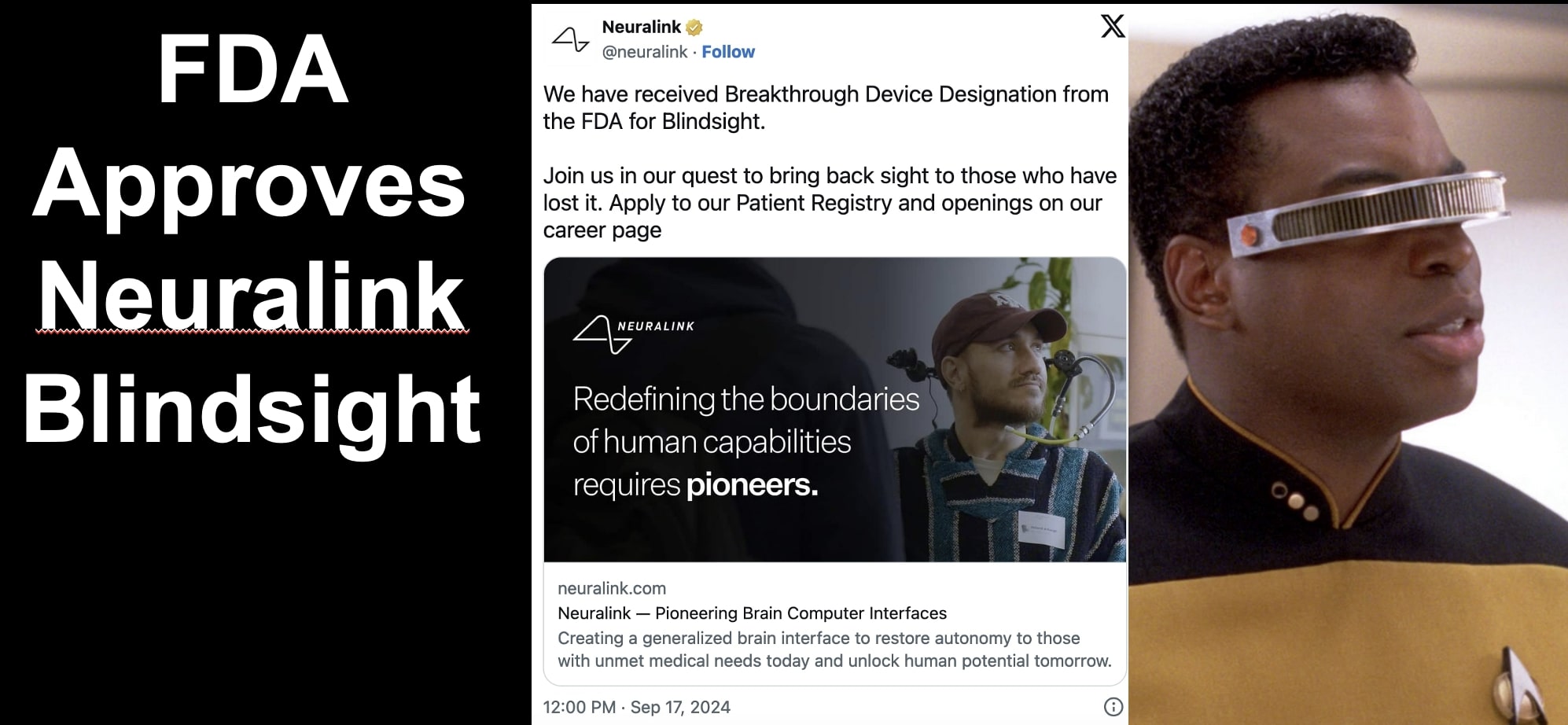
Elon Musk's Neuralink announced its AI-powered Blindsight brain implant, designed to restore vision even for those blind from birth or without optic nerves, has received the FDA's 'breakthrough device' designation. The device is experimental, with no reported harm, and aims to enable future human trials.[AI generated]

















































)