The information displayed in the AIM should not be reported as representing the official views of the OECD or of its member countries.
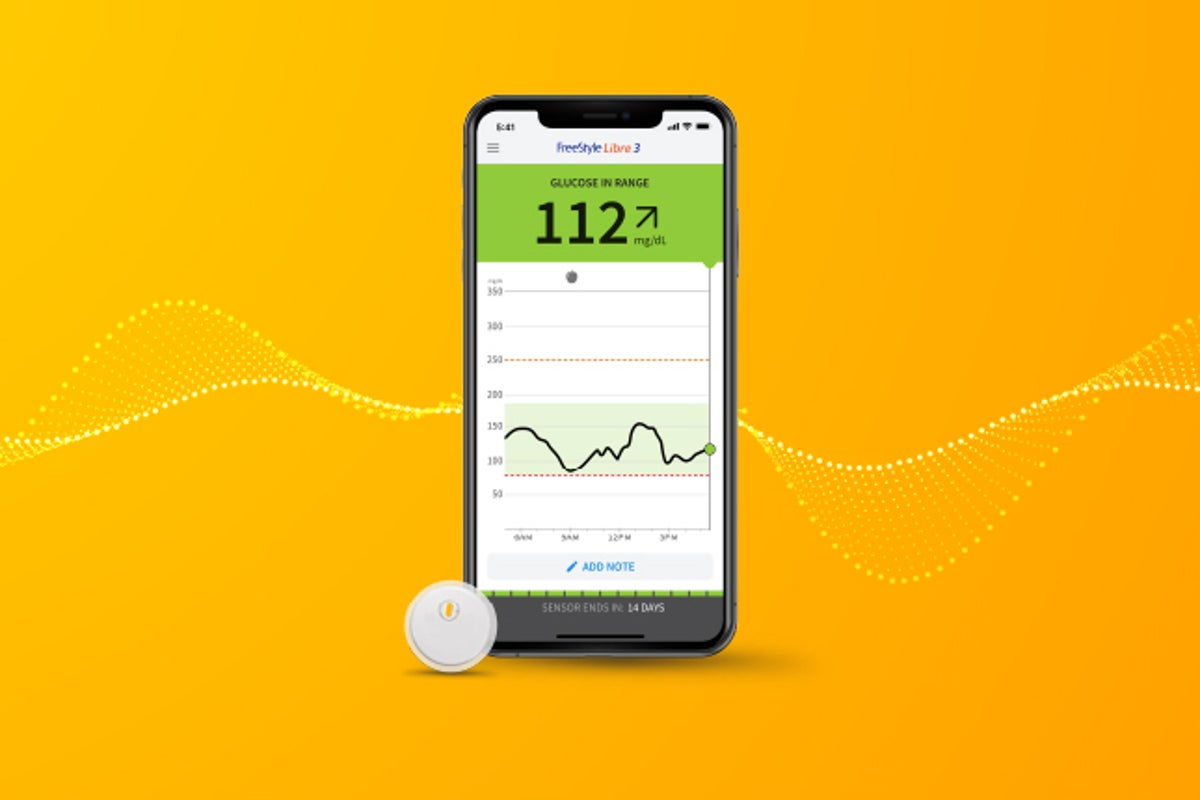
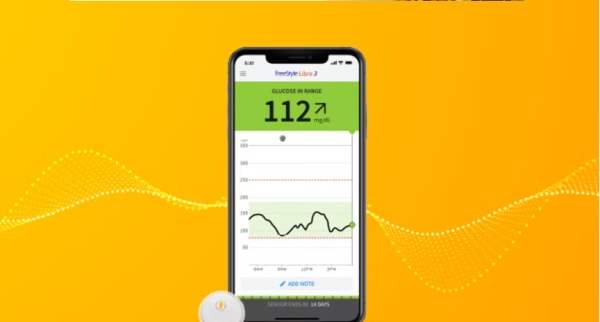
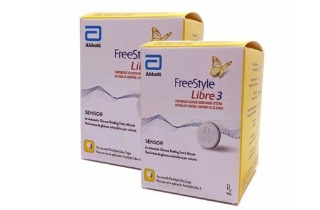
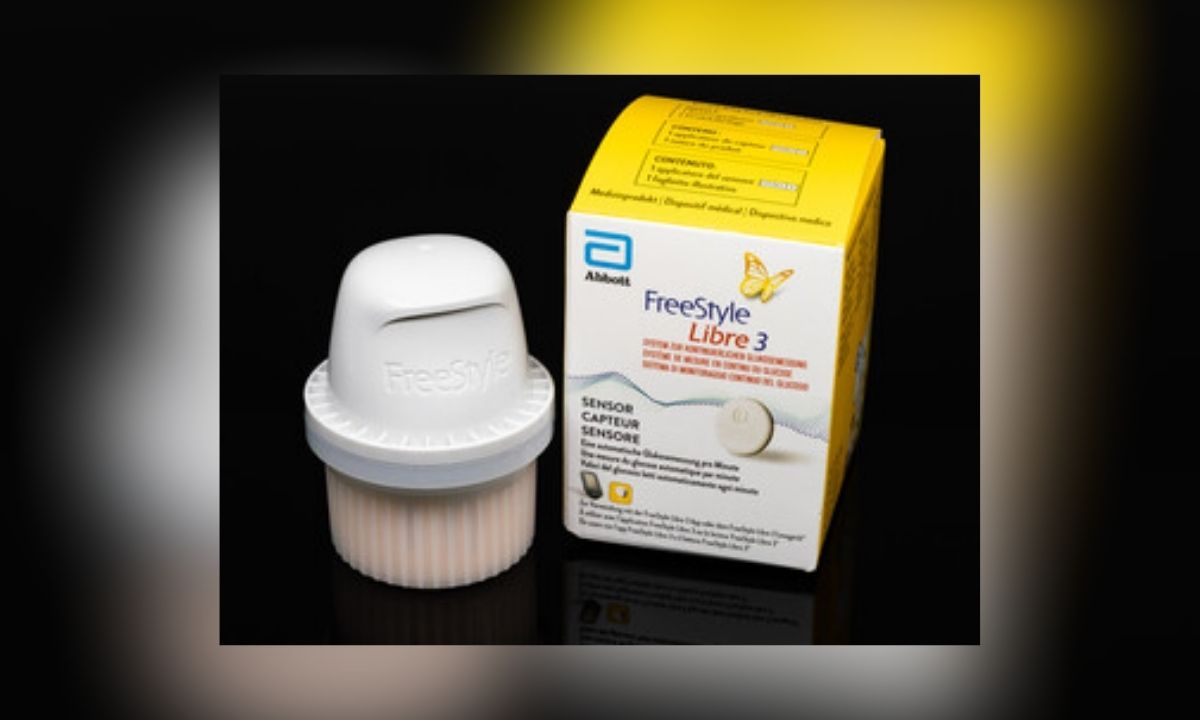
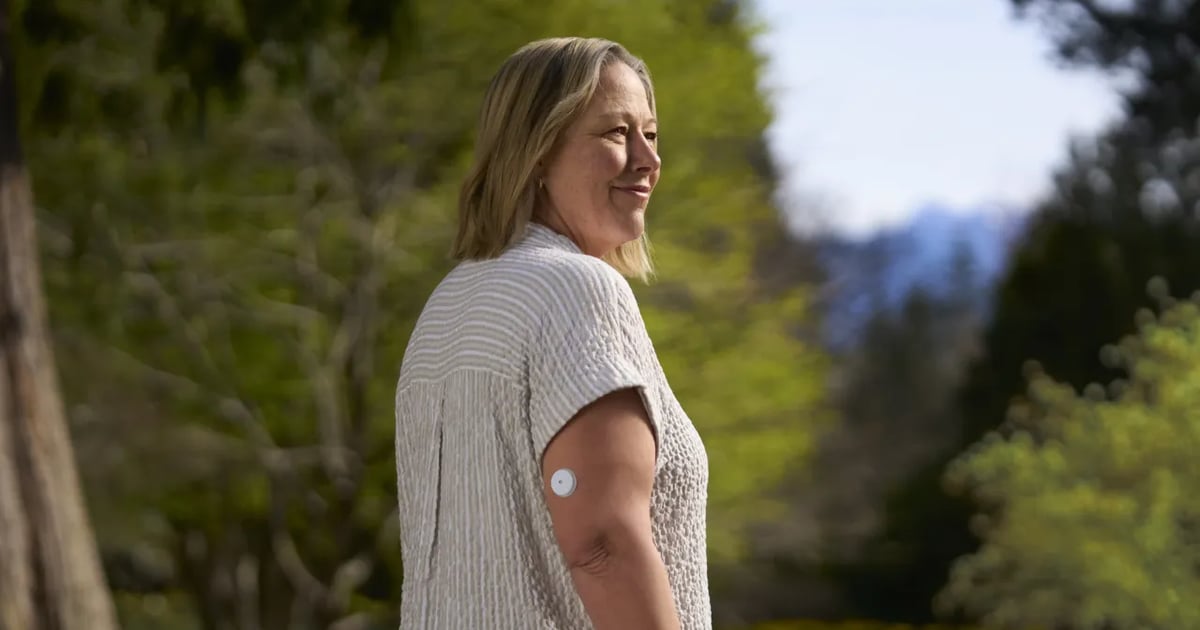
Abbott recalled about 3 million FreeStyle Libre 3 glucose sensors in the U.S. after a manufacturing defect caused AI-driven devices to give false low blood sugar readings. The malfunction led to 736 severe adverse events and seven deaths worldwide, prompting device corrections and replacement efforts.[AI generated]





































:max_bytes(150000):strip_icc()/Health-cgm-recall-01e2225abe9a4583a63c2bfefe68bd8f.png)